
Kalyan Nagar, Bangalore, India - 560043
4
Details verified of Malus Coaching Classes✕
 Identity
Identity
 Education
Education
Know how UrbanPro verifies Tutor details
Identity is verified based on matching the details uploaded by the Tutor with government databases.

No. 302, 5th D Main Road , 2nd Floor, 3rd Cross, HRBR layout, 2nd Block
Kalyan Nagar, Bangalore, India - 560043
Landmark: #302, B Main Road, 3rd cross, HRBR-II Block, Kalyan Nagar, Bangalore-560043
![]() Phone Verified
Phone Verified
![]() Email Verified
Email Verified
Report this Profile
Is this listing inaccurate or duplicate? Any other problem?
Please tell us about the problem and we will fix it.
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
ICSE, State, NIOS, CBSE
Subjects taught
Tamil, Commercial Applications, Home Science, Chemistry, Accountancy, Art, Physics, Marathi, Science, Environmental Applications, Mathematics, History and Civics, Social Science, Hindi, Sanskrit, Geography, English, Kannada, Urdu, EVS, Economic Application, Spanish, Commercial Studies, Biology, Computer Application, Economics, German, Telugu, English Literature, Bengali, French
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
ICSE, State, NIOS, CBSE
Subjects taught
Kannada, Manipuri, Punjabi, Geography, Information and Comunication Technology, Urdu, Science, Commercial Studies, Computer Practices, Chemistry, Telugu, Environmental Applications, Tamil, Physics, English, Bengali, Computer Application, Elements of business, Japanese, History and Civics, Gujarati, Mathematics, Marathi, French, Economic Application, Accountancy, Commercial Applications, Spanish, EVS, English Literature, Art, Biology, Social Science, Oriya, Malayalam, German, Economics, Home Science, Hindi, Sanskrit
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
NIOS, State, CBSE, Cambridge Assessment International Education (CAIE), ICSE
Subjects taught
German, Chemistry, Kannada, Computer Science, Science, Physics, Sanskrit, EVS, Biology, History, Computers, Mathematics, Social science, Geography, Hindi, English
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
NIOS, State, CBSE, Cambridge Assessment International Education (CAIE), ICSE
Subjects taught
Chemistry, Science, Geography, Computers, Mathematics, Hindi, History, Social science, Kannada, Computer Science, German, Physics, Biology, English, Sanskrit, EVS
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
NIOS, State, CBSE, Cambridge Assessment International Education (CAIE), ICSE
Subjects taught
Biology, German, Computer Science, History, Geography, Mathematics, Social science, Science, Hindi, Physics, Chemistry, EVS, English, Computers, Sanskrit, Kannada
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Coaching provided for
CA Final Group 2, CA Foundation, IPCC Group 1, IPCC Group 2, CA Final Group 1
CA_IPCC_Subjects_Group II
Auditing and Assurance, Information Technology and Strategic Management, Advanced Accounting
CA_IPCC_Subjects_Group I
Accounting, Cost Accounting and Financial Management, Taxation, Business Laws, Ethics and Communication
CA_CPT_Subjects
Fundamentals OF Accounting, General English, Quantitative aptitude, General Economics
CA Final_subjects_Group I
Advanced Auditing and Professional Ethics, Financial Reporting, Strategic Financial Management, Corporate and Allied Laws
CA_Final_Subjects_Group II
Advanced Management Accounting, Direct Tax Laws, Indirect Tax Laws, Information Systems Control and Audit
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Engineering Entrance Exams
GATE Coaching Classes, BITSAT Coaching Classes, IIT JEE Coaching Classes
Type of class
Crash Course, Regular Classes
IIT JEE Coaching
IIT JEE Advanced Coaching, IIT JEE Foundation Course, IIT JEE Mains Coaching, IIT JEE Crash Course, IIT JEE Integrated Coaching
IIT-JEE Subjects
Physics , Chemistry , Maths
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Teaches for following exams
AIPMT Coaching, Others, AIIMS Coaching
Type of class
Crash Course, Regular Classes
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
State, CBSE, ISC/ICSE, IGCSE
Subjects taught
Geography, Electronics, Computer Science, Environmental Management, Sociology, History, Accountancy, Political Science, Chemistry, Economics, Statistics, Home Science, German, Biotechnology, Sanskrit, Mathematics, Physics, Hindi, EVS, English Literature, Social science, Geology, Biology, Commerce, English, Accounts, Business Studies, Kannada
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
State, CBSE, ISC/ICSE, NIOS, IGCSE
Subjects taught
Hindi, English Literature, Home Science, Mathematics, Biology, Accountancy, Chemistry, Environmental Management, Business Studies, Commerce, Social science, Geology, History, Sociology, Statistics, English, EVS, Electronics, German, Physics, Accounts, Political Science, Sanskrit, Computer Science, Biotechnology, Kannada, Geography, Economics
1. Which school boards of of Class 10 do you teach for?
ICSE, State, NIOS and others
2. Which classes do you teach?
We teach CA Coaching, Class 10 Tuition, Class 11 Tuition, Class 12 Tuition, Class 6 Tuition, Class 7 Tuition, Class 8 Tuition, Class 9 Tuition, Engineering Entrance Coaching and Medical Entrance Coaching Classes
3. Do you provide a demo class?
Yes, We provide a free demo class.
4. Where are you located?
We are located in Kalyan Nagar, Bangalore.
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
(a) When silicon reacts with methyl chloride in the presence of copper (catalyst) and at a temperature of about 537 K, a class of organosilicon polymers called methyl-substituted chlorosilanes are formed. Examples, 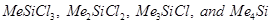
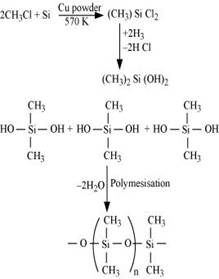
(b) When silicon dioxide (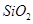 ) is heated with hydrogen fluoride (HF), it forms silicon tetrafluoride (
) is heated with hydrogen fluoride (HF), it forms silicon tetrafluoride (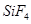 ). Usually, the Si-O bond is a strong bond and it resists any attack by halogens and most acids, even at a high temperature. However, it is attacked by HF.
). Usually, the Si-O bond is a strong bond and it resists any attack by halogens and most acids, even at a high temperature. However, it is attacked by HF.
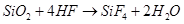
The 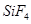 formed in this reaction can further react with HF to form hydrofluorosilicic acid.
formed in this reaction can further react with HF to form hydrofluorosilicic acid.
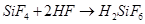
(c) When CO reacts with ZnO, it reduces ZnO to Zn. CO acts as a reducing agent.
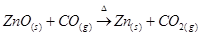
(d) When hydrated alumina is added to sodium hydroxide, the former dissolves in the latter because of the formation of sodium meta-aluminate.
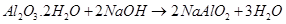
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
The elements of group 14 have 4 electron’s in the outermost shell i.e valence shell hence the oxidation state of the group is +4
As the result of the inert pair effect the lower oxidation state becomes more and more stable and opposite things happen on the other hand the highest oxidation state becomes less and less stable
Therefore the group has oxidation state from +4 to +2
C (carbon) = oxidation state +4
Si (silicon) = oxidation state +4
Sn, Pb (lead) = oxidation state +2,+4
Answered on 05/01/2019 Learn CBSE
We now turn to the synthesis of glucose from noncarbohydrate precursors, a process called gluconeogenesis.
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
ICSE, State, NIOS, CBSE
Subjects taught
Tamil, Commercial Applications, Home Science, Chemistry, Accountancy, Art, Physics, Marathi, Science, Environmental Applications, Mathematics, History and Civics, Social Science, Hindi, Sanskrit, Geography, English, Kannada, Urdu, EVS, Economic Application, Spanish, Commercial Studies, Biology, Computer Application, Economics, German, Telugu, English Literature, Bengali, French
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
ICSE, State, NIOS, CBSE
Subjects taught
Kannada, Manipuri, Punjabi, Geography, Information and Comunication Technology, Urdu, Science, Commercial Studies, Computer Practices, Chemistry, Telugu, Environmental Applications, Tamil, Physics, English, Bengali, Computer Application, Elements of business, Japanese, History and Civics, Gujarati, Mathematics, Marathi, French, Economic Application, Accountancy, Commercial Applications, Spanish, EVS, English Literature, Art, Biology, Social Science, Oriya, Malayalam, German, Economics, Home Science, Hindi, Sanskrit
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
NIOS, State, CBSE, Cambridge Assessment International Education (CAIE), ICSE
Subjects taught
German, Chemistry, Kannada, Computer Science, Science, Physics, Sanskrit, EVS, Biology, History, Computers, Mathematics, Social science, Geography, Hindi, English
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
NIOS, State, CBSE, Cambridge Assessment International Education (CAIE), ICSE
Subjects taught
Chemistry, Science, Geography, Computers, Mathematics, Hindi, History, Social science, Kannada, Computer Science, German, Physics, Biology, English, Sanskrit, EVS
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
NIOS, State, CBSE, Cambridge Assessment International Education (CAIE), ICSE
Subjects taught
Biology, German, Computer Science, History, Geography, Mathematics, Social science, Science, Hindi, Physics, Chemistry, EVS, English, Computers, Sanskrit, Kannada
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Coaching provided for
CA Final Group 2, CA Foundation, IPCC Group 1, IPCC Group 2, CA Final Group 1
CA_IPCC_Subjects_Group II
Auditing and Assurance, Information Technology and Strategic Management, Advanced Accounting
CA_IPCC_Subjects_Group I
Accounting, Cost Accounting and Financial Management, Taxation, Business Laws, Ethics and Communication
CA_CPT_Subjects
Fundamentals OF Accounting, General English, Quantitative aptitude, General Economics
CA Final_subjects_Group I
Advanced Auditing and Professional Ethics, Financial Reporting, Strategic Financial Management, Corporate and Allied Laws
CA_Final_Subjects_Group II
Advanced Management Accounting, Direct Tax Laws, Indirect Tax Laws, Information Systems Control and Audit
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Engineering Entrance Exams
GATE Coaching Classes, BITSAT Coaching Classes, IIT JEE Coaching Classes
Type of class
Crash Course, Regular Classes
IIT JEE Coaching
IIT JEE Advanced Coaching, IIT JEE Foundation Course, IIT JEE Mains Coaching, IIT JEE Crash Course, IIT JEE Integrated Coaching
IIT-JEE Subjects
Physics , Chemistry , Maths
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Teaches for following exams
AIPMT Coaching, Others, AIIMS Coaching
Type of class
Crash Course, Regular Classes
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
State, CBSE, ISC/ICSE, IGCSE
Subjects taught
Geography, Electronics, Computer Science, Environmental Management, Sociology, History, Accountancy, Political Science, Chemistry, Economics, Statistics, Home Science, German, Biotechnology, Sanskrit, Mathematics, Physics, Hindi, EVS, English Literature, Social science, Geology, Biology, Commerce, English, Accounts, Business Studies, Kannada
Class Location
![]() Online (video chat via skype, google hangout etc)
Online (video chat via skype, google hangout etc)
![]() At the Institute
At the Institute
Board
State, CBSE, ISC/ICSE, NIOS, IGCSE
Subjects taught
Hindi, English Literature, Home Science, Mathematics, Biology, Accountancy, Chemistry, Environmental Management, Business Studies, Commerce, Social science, Geology, History, Sociology, Statistics, English, EVS, Electronics, German, Physics, Accounts, Political Science, Sanskrit, Computer Science, Biotechnology, Kannada, Geography, Economics
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
(a) When silicon reacts with methyl chloride in the presence of copper (catalyst) and at a temperature of about 537 K, a class of organosilicon polymers called methyl-substituted chlorosilanes are formed. Examples, 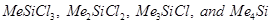

(b) When silicon dioxide (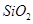 ) is heated with hydrogen fluoride (HF), it forms silicon tetrafluoride (
) is heated with hydrogen fluoride (HF), it forms silicon tetrafluoride (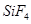 ). Usually, the Si-O bond is a strong bond and it resists any attack by halogens and most acids, even at a high temperature. However, it is attacked by HF.
). Usually, the Si-O bond is a strong bond and it resists any attack by halogens and most acids, even at a high temperature. However, it is attacked by HF.
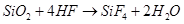
The 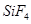 formed in this reaction can further react with HF to form hydrofluorosilicic acid.
formed in this reaction can further react with HF to form hydrofluorosilicic acid.
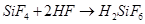
(c) When CO reacts with ZnO, it reduces ZnO to Zn. CO acts as a reducing agent.
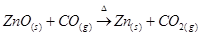
(d) When hydrated alumina is added to sodium hydroxide, the former dissolves in the latter because of the formation of sodium meta-aluminate.
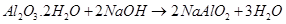
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements
The elements of group 14 have 4 electron’s in the outermost shell i.e valence shell hence the oxidation state of the group is +4
As the result of the inert pair effect the lower oxidation state becomes more and more stable and opposite things happen on the other hand the highest oxidation state becomes less and less stable
Therefore the group has oxidation state from +4 to +2
C (carbon) = oxidation state +4
Si (silicon) = oxidation state +4
Sn, Pb (lead) = oxidation state +2,+4
Answered on 05/01/2019 Learn CBSE
We now turn to the synthesis of glucose from noncarbohydrate precursors, a process called gluconeogenesis.
Answered on 05/01/2019 Learn CBSE/Class 11/Science/Chemistry/Unit 11-Some p -Block Elements

Share this Profile
Also have a look at
Reply to 's review
Enter your reply*
Your reply has been successfully submitted.
Certified
The Certified badge indicates that the Tutor has received good amount of positive feedback from Students.