The acid dissociation constant Ka is a quantitative measure of the strength of an acid in the solution.
Pka: A larger Ka value means a stronger tendency to dissociate a proton, and thus, it means the substance is a stronger acid.
Also, Ka is the acid ionization constant, the equilibrium constant for chemical reactions involving weak acids in acqueous solution. The numerical value of Ka is used to predict the extent of acid dissociation.
pka = -log(Ka)
This equation is called Henderson-Hasselbalch equation.
The lower the pka, the stronger the acid.
pka=ph+ log(acid/base)
Let us now study an example to determine which compound has greater pka value.
as F) is very electron withdrawing
hence, it has greater acid strength and lower pka value.
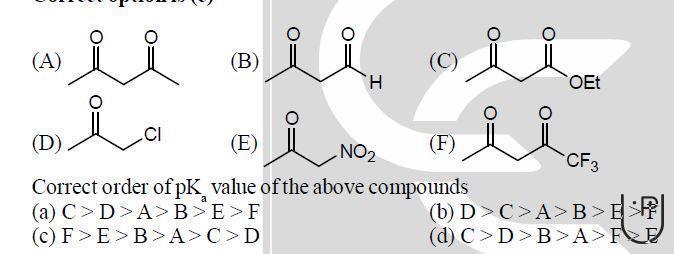
ANSWER: B)


 0
0

